Scientists develop antibodies that block Epstein Barr Virus in humanized mice
Fred Hutch Cancer Center scientists have developed genetically human monoclonal antibodies that block Epstein Barr Virus from binding to and entering immune cells, and one of those antibodies prevented infection when tested in mice with human immune systems.
Epstein Barr Virus targets: gp350 and gp42
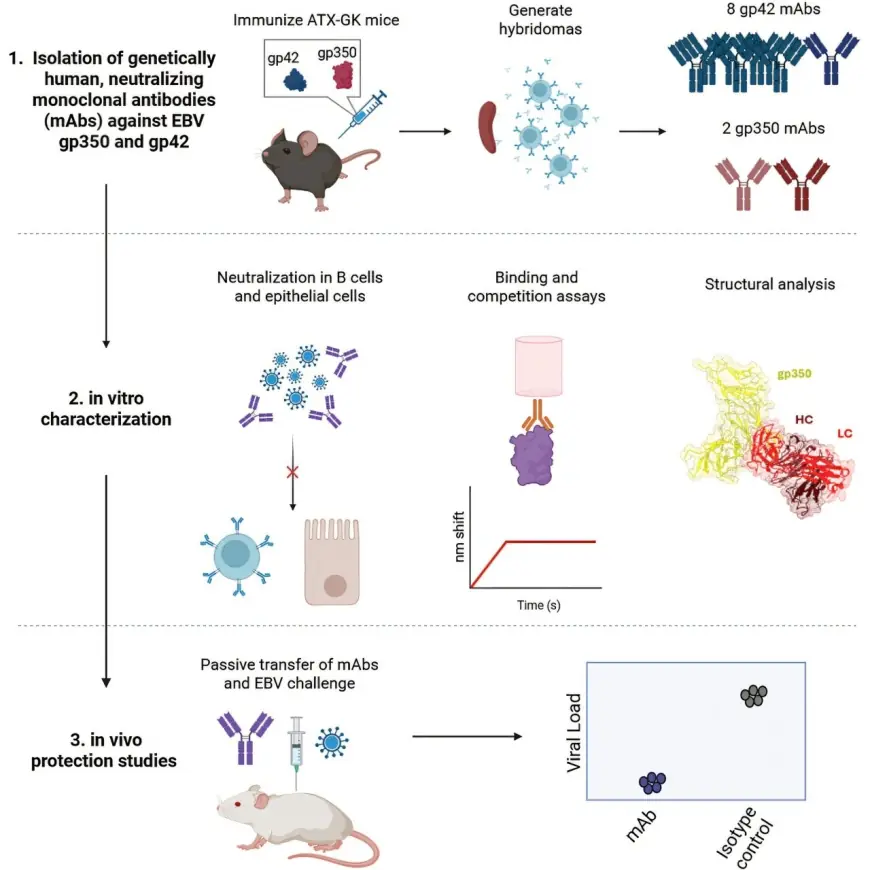
The research team focused on two viral surface proteins: gp350, which helps the virus bind to cell receptors, and gp42, which facilitates entry by fusion. Using a mouse model engineered with human antibody genes, the scientists produced a total of ten monoclonal antibodies—two directed at gp350 and eight aimed at gp42. In the study’s final step, one monoclonal antibody against gp42 successfully prevented EBV infection in humanized mice, while another antibody against gp350 provided partial protection. The work was published in Cell Reports Medicine (DOI: 10. 1016/j. xcrm. 2026. 102618).
How the antibodies worked in mice with human immune systems
Researchers used mice with human antibody genes and mice with human immune systems to test whether the new antibodies could stop the virus from attaching and entering B cells. The team identified sites of vulnerability on gp350 and gp42 that the antibodies target, and laboratory challenge experiments showed that blocking those sites can halt infection. "Finding human antibodies that block Epstein Barr virus from infecting our immune cells has been particularly challenging because, unlike other viruses, EBV finds a way to bind to nearly every one of our B cells, " said Andrew McGuire, Ph. D., a biochemist and cellular biologist in the Vaccine and Infectious Disease Division at Fred Hutch. The study validated an approach for discovering protective antibodies that could inform vaccine design for EBV and other pathogens.
Implications for transplant patients and separate findings on microplastics
The work has specific relevance for people who undergo immune suppression. More than 128, 000 people in the U. S. undergo solid organ and bone marrow transplant annually, and post-transplant lymphoproliferative disorders (PTLD), most of which are EBV-associated lymphomas, remain a frequent cause of morbidity and mortality after organ transplantation, noted Rachel Bender Ignacio, MD, MPH, an associate professor and infectious disease physician at Fred Hutch and University of Washington School of Medicine. Because there are currently no targeted therapies to prevent EBV infection or reactivation in immunosuppressed transplant patients, antibodies that block viral binding and entry could offer a new preventive tool.
Separately highlighted in related coverage, a recent study identified microplastics in 90% of prostate cancer samples, with tumors containing 2. 5 times more plastic fragments than noncancerous prostate tissue. Researchers noted that more evidence is required to confirm any causal link between microplastic exposure and malignancy.
"Not only did we identify important antibodies against Epstein Barr virus, but we also validated an innovative a new approach for discovering protective antibodies against other pathogens, " said Crystal Chhan, a pathobiology Ph. D. student in the McGuire Lab, reflecting on how the project yielded unexpected discoveries while pursuing human monoclonal antibodies that avoid triggering anti-drug responses.
The team’s next confirmed outcome in the published work was mapping the sites of vulnerability on gp350 and gp42; the study notes those sites could be useful in future vaccine development and in designing therapies to protect high-risk patients. Further laboratory and preclinical work tied to those vulnerability sites is the immediate path the paper outlines for translating these antibody findings into vaccine or therapeutic strategies.