BMI-Based Semaglutide Guidelines Risk Excluding Vulnerable Indian Population
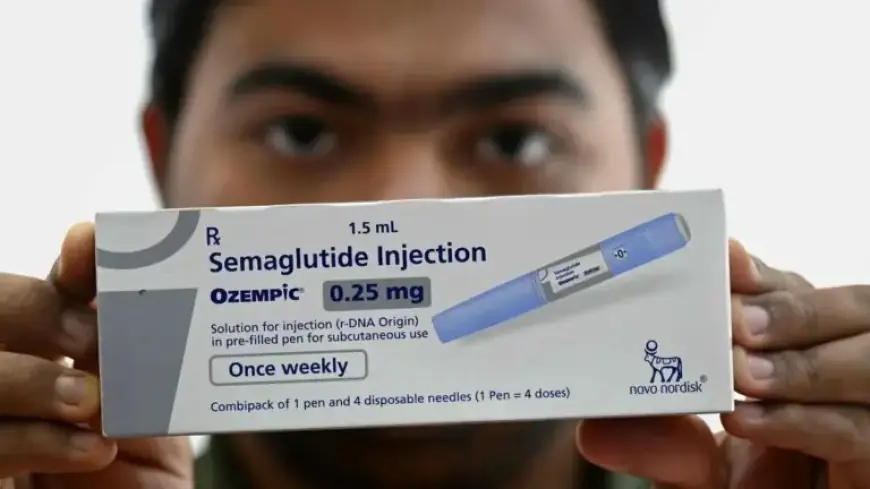
The SELECT cardiovascular outcomes trial showed once-weekly semaglutide lowered major cardiovascular events by 20 percent. The benefit appeared in adults with a body mass index of 27 or higher and established cardiovascular disease, but without diabetes.
Trial design and main findings
The trial was led by A. Michael Lincoff and published in The New England Journal of Medicine in 2023. It enrolled 17,604 participants and followed them for nearly 40 months. Investigators reported a 20% reduction in major cardiovascular events with semaglutide compared with placebo.
Eligibility criterion and its limits
The study applied a BMI threshold of 27 or greater to define eligibility. That threshold excluded people with lower body weight. The trial therefore focused on adults with higher BMI and pre-existing cardiovascular disease, but without diabetes.
Concerns for Indian patients
Experts warn the BMI cutoff may not match risk patterns seen in India. Aditi Kantipuly, a physician training in preventive medicine and public health resident at McGill University and a visiting scholar at the University of New Mexico, highlighted this concern. She noted many Indian patients develop cardiovascular disease at lower body weights.
Policy implications
Critics say BMI-Based Semaglutide Guidelines could carry a risk of excluding vulnerable Indian population. They call for locally adapted thresholds that reflect regional risk profiles. Adjusted guidance could better identify high-risk patients who might benefit from treatment.
Next steps
Further evaluation of semaglutide eligibility in South Asian cohorts is needed. Local data should inform guideline updates. Policymakers and clinicians must consider population differences when applying international trial criteria.
Published April 08, 2026 02:37 pm IST.