Exploring NTSR1 G Protein Subtype Dynamics and Promiscuity
Research into G protein dynamics has gained significant traction, especially concerning the NTSR1 G protein subtype. This article summarizes the key procedures and findings related to the expression, purification, and complex formation of NTSR1 with G proteins.
Expression and Purification of G Protein Subtypes
The G protein heterotrimer was expressed in Tni cells using a baculovirus system. Following expression, the cells were snap-frozen and prepared for purification. The purification process involved suspension in a lysis buffer, centrifugation, and washing steps to yield the desired protein.
- Expression System: Tni cells
- Purification Buffer: 20 mM HEPES, 1 mM EDTA, 5% glycerol
Subsequent to the initial purification, the G protein was solubilized and concentrated through advanced chromatography techniques, including the use of Ni-NTA and size-exclusion methods. Final samples were snap-frozen for later analysis.
Purification Process Overview
The overall procedure for purifying G proteins included the following steps:
- Resuspension in lysis buffer
- Ultracentrifugation of cell lysates
- Solubilization in a specific buffer
- Affinity and size-exclusion chromatography
- Concentration and freezing for later use
Complex Formation of NTSR1 with G Protein
The formation of the NTSR1–G protein complex began by incubating NTSR1 with a G protein in significant excess. Following the initial incubation, apyrase and a protease were added to enhance complex stability. The formed complexes were subjected to further purification steps using specific resin and chromatography.
- Incubation Time: 1 hour on ice
- Cleavage Agent: HRV 3C protease
Following complex formation, additional purification techniques were applied to refine the NTSR1–G protein complex. These included affinity chromatography to eliminate non-specific protein interactions and size-exclusion chromatography for better separation based on size.
Advanced Analytical Techniques
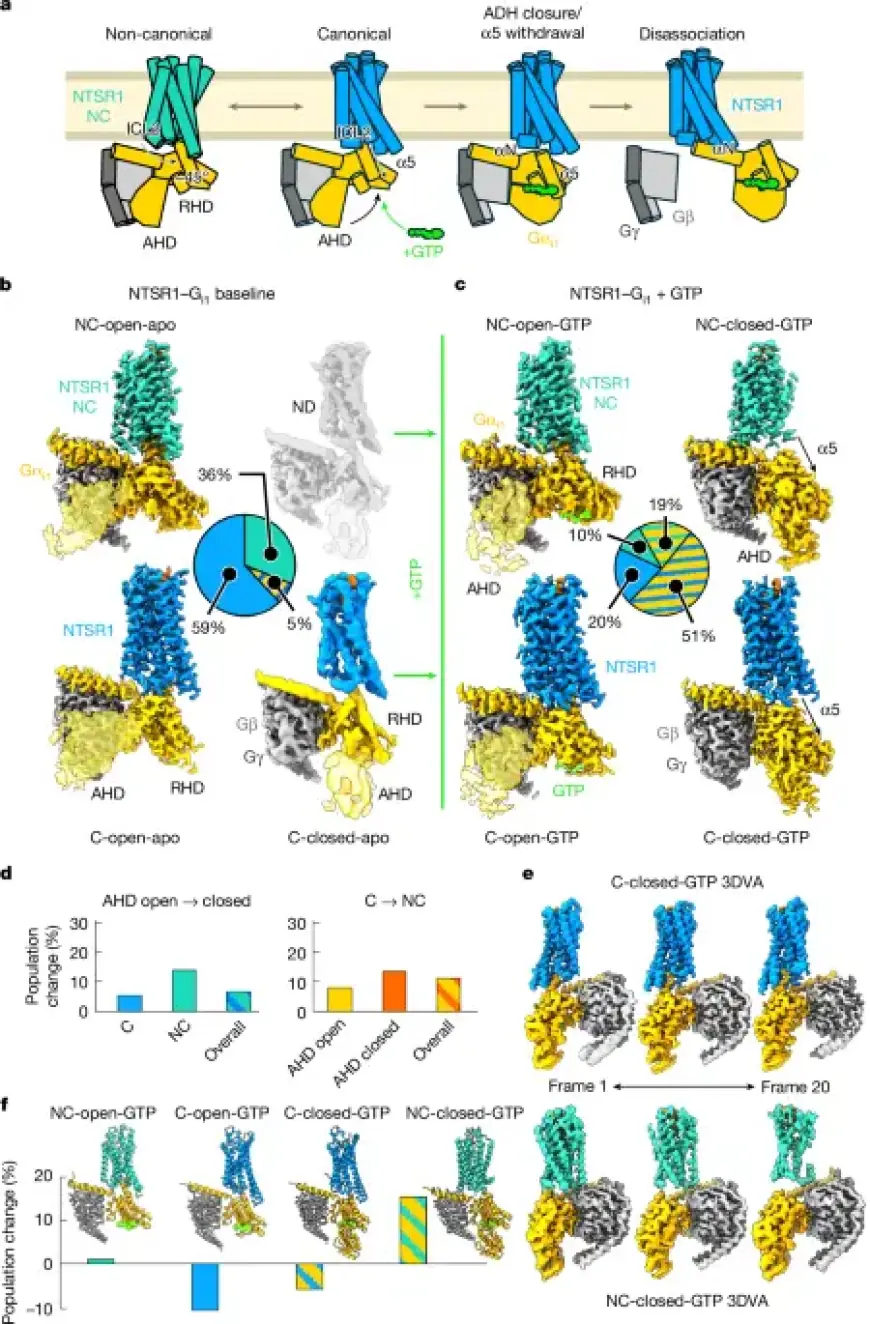
Time-resolved cryo-electron microscopy (cryo-EM) was employed to analyze the complex formed by NTSR1 and G proteins. Sample preparation involved careful grid freezing, and data collection utilized advanced electron microscopy equipment.
- Grid Type: UltrAufoil R1.2/1.3
- Equipment: G4 Titan Krios with K3 detector
Explore the implications of this research for understanding G protein dynamics and their potential applications in biochemical research and drug design. This study highlights the significance of NTSR1 receptor and its interactions with G protein subtypes, paving the way for future investigations.
Conclusion
The exploration of NTSR1 G protein subtype dynamics provides valuable insights into G protein signaling pathways. The methodologies outlined here contribute to a better understanding of receptor interactions, essential for future therapeutic developments.